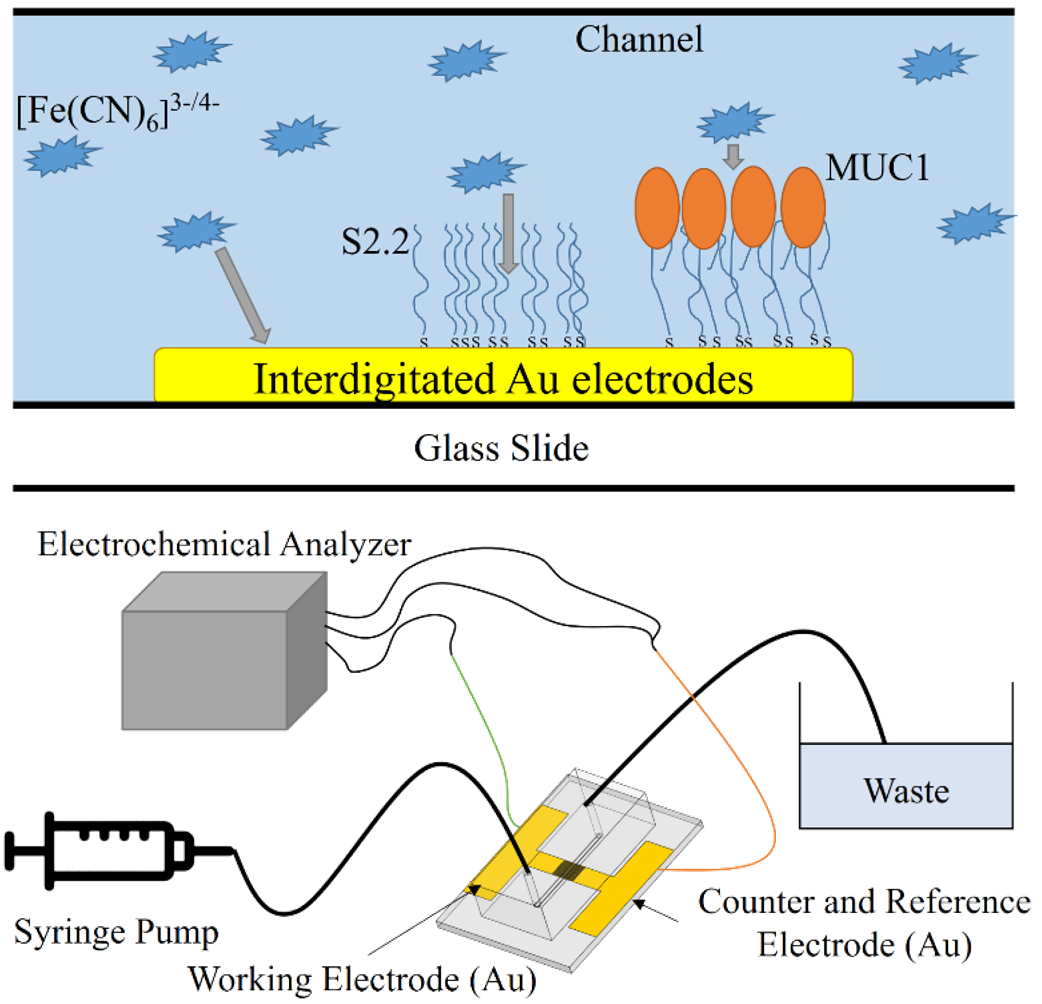
This study serves as a major extension for my previous research Impedimetric Microfluidic Chip for MUC1 Aptasensing. Similar to the previous one, microfluidic chips are fabricated for impedimetric detection of tumor marker MUC1. Moreover, I integrated an interdigitated array electrode (IDA electrode) into the chip, and realized real-time detection for the aptasensor. In-depth investigation of the relationship between electrochemical properties and microfluidic conditions are also carried out. This system possesses several advantages, such as the highly sensitive characteristic of IDA electrodes, real-time detection, low sample usage, label-free detection using EIS, and miniaturized volume using a microfluidic chip.
Introduction
MUC1 is a surface glycoprotein which over-expresses in several types of cancer cells, making it an ideal marker for cancer detection. For MUC1 recognition, the S2.2 aptamer is a 25mer ssDNA selected in vitro and can affinitively and specifically bind to certain motifs within the MUC1 protein. Electrochemical impedance spectroscopy (EIS) has been proven as an effective method for ultrasensitive MUC1 aptasensing and cell detection [1]. Despite the high sensitivity and selectivity of EIS and the S2.2 aptamer, long reaction times and large sample volumes have hindered EIS biosensors for realistic bioanalysis.
The integration of microfluidics with EIS has a large potential for lowering the amount of usage during reactions and to meet real-time, portable, large-scale and high-throughput requirements. Though up to date, there hasn’t yet been studies regarding real-time impedimetric aptasensing to the best of our knowledge. Thus, this research is dedicated to develop a real-time microfluidic impedance aptasensing platform for affinitive and selective detection of MUC1.
Related Publications
- C.-Y. Lai, J.-H. Weng, L.-C. Chen, Real-time impedimetric MUC1 aptasensor using microfluidic symmetric Au electrodes, The Twenty Second International Conference on Miniaturized Systems for Chemistry and Life Sciences (µTAS), (2018).
[poster pdf] - J.-H. Weng, C.-Y. Lai, L.-C. Chen, Microfluidic amperometry with two symmetric Au microelectrodes under one-way and shuttle flow conditions, Electrochimica Acta, 297 (2019) 118-128.
https://doi.org/10.1016/j.electacta.2018.11.128